|
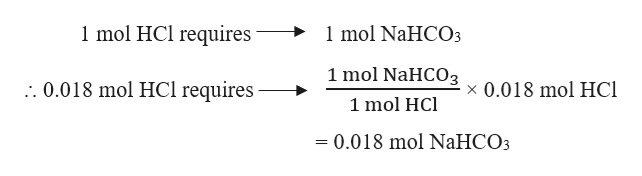
And that retested So would be 82.2 divided by 1.539 is the mass of our weak acid is equivalent to 53.4g, implying that the mass of our country base Is equivalent to 28.8 g. So we know the massive A zero point factory. So now we can substitute this ratio and determine the total mess so we can substitute. And were given information that the mass of our contrary base plus the mass Our rig acid is equivalent to 82.2 g. So, Molar mass of NaHCO3 Molar mass of 1 Sodium (Na) atom + Molar mass of 1 Hydrogen (H) atom + Molar mass of 1 Carbon (C) atom + Molar mass of 3 Oxygen (O) atoms. So the ratios of the masses By rearranging is equivalent to Times one. And we can simplify this down a bit 70 mass of trying to get bess Times 84 divided by the mass Folic Aid Times 106 Equals 0.4-7. And then we have the mass of are weak acid And this has a molar mass of 84g promote and this is equivalent to once again this so now we can move all of our factors over to the other side So we can multiply by 84 and by 106 Multiplied by 84 and 106. So it has a molar mass of about 106 g per mole. So can you get based So the contact base is has a molar mass. So the number of moles is essentially the mass of certain components of the mass of h minus over The molar mass of 18. This has number of moles of contact base over that of our weak acid. So let's first determine this in terms of technically we can use molds as well since it's one leaders. And this essentially gives information of the ratios of the concentrations of our conscious base and are weak acids. So we'll be using the PK2 which is equivalent to 10.32. So it's important to remember that carbonic acid is a die protic acid. So I'm going to write this as a tu minus over H a minus. And since this is a buffer system, we can apply the Henderson Hasselbach equation. Molar mass of NaHCO3 84 Convert between NaHCO3 weight and moles Convert between NaHCO3 weight and moles. 
So we're given information that the ph is equivalent to 9.95. So essentially we have a buffer system here. So this is an example of a weak acid while our carbon, it is an example of a conjugate base. It can be calculated by adding the invididual molar mass of every atom that are composing the molecule (CH4 : 1 atom of C -> molar mass 12.011 g/mol + 4 atom of H -> molar mass 1.008 g/mol -> molar mass of CH4 12.011+41.00816. Sana has been working at Whatsinsight since 2020 as a content writer.So here we give an example of a certain mixture and we have bicarbonate and carbonate. Related Links CO 2 Lewis Structure and Molecular Geometry HCN Lewis Structure| Step By Step Construction SO 2 (Sulfur Dioxide) Lewis structure N 2O Lewis Structure| Laughing Gas What is the Molar Mass of Nitrogen? What is NaHCO3 in molar mass The molar mass of NaHCO3 84.00661 g/mol What is the molar mass of NaHCO3 the answer is 105.9886 g/mole molecular weight calculation is. Since CH 3COOH forms ice-like crystals below room temperature (16.6 ☌ or 61.9 ☏), it is also called “glacial acetic acid.” Important Links The name glacial acetic acid comes from the German name Eisessig (ice vinegar).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
